ABSTRACT
Background and Objective: The cultivation of Vegetables in the world is facing a shortage of water so that the farmers are forced to use sewage wastewater for cultivation in underdeveloped countries. Therefore, the present study was an attempt to examine the toxicity level of accumulated heavy metals in the vegetables irrigated with sewage water and treated sewage water. The concentration-dependent changes in toxicity of ethanolic leaf and fruit extracts of Phaseolus vulgaris treated with wastewater in Zebrafish were analysed in this study. Materials and Methods: For the experiment, finely ground powders of leaves and fruits of Phaseolus vulgaris were extracted with ethanol. Using different concentrations of these extracts, a toxicity test was done with Danio rerio as per the OECD guidelines 203. Results: Using AAS, heavy metals like lead and manganese were found in higher concentrations in untreated wastewater than in distilled water and treated wastewater. The results indicated that ethanolic leaf extracts of treated wastewater irrigated Phaseolus vulgaris does not induce toxicity when used at a dose below 400 mg L–1. Leaf extracts of Phaseolus vulgaris grown with wastewater showed the lowest and highest mortality at 100 and 400 mg L–1, respectively, when compared to other plant extracts. Histopathological variations were also observed in the fishes exposed to the lethal concentrations of plant extracts. Statistical evaluation of the correlation between concentration and mortality percentage was carried out using SPSS. Conclusion: The present study revealed that the leaf and fruit extracts of Phaseolus vulgaris grown with untreated wastewater were more toxic to Danio rerio than other extracts used in the experiment.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2022.24.31
URL: https://scialert.net/abstract/?doi=ajps.2022.24.31
INTRODUCTION
Phaseolus vulgaris L. or common bean, belonging to the legume family Fabaceae, is widely used as a vegetable in different parts of the world1. Depending on the variety, common beans can be bushy (dwarf beans), vine-like, or climbing (pole beans). The plant grows well at temperatures ranging from 15-20°C. The optimal growth conditions to reduce the risk of bacterial and fungal disease include 350-500 mm of rain per season along with low relative humidity. Low temperatures slow down plant growth while high temperatures accelerate it as they are warm-season crops. The common bean prefers rich, moist soil that is loose to mildly compress with good physical properties and a pH of 5.5-6.5 and is flat or rolling with good drainage. The seedbed should be irrigated before planting to ensure uniform moisture to the soil. Soil should be watered lightly after planting to moisten it and dampen the crop2.
Irrigation is essential for assuring sufficient soil moisture for better plant growth. Water stress can affect the quality of the yield of flowers, fruits and vegetables. The need for irrigation is influenced by factors like timing and amount of rainfall during the season, the water-holding capacity of soil and crop's water requirement. Soil structure determines the movement of air, water and roots through the soil. Both the crops and the soils on which irrigation water can be distributed must be compatible. The physical characteristics, atmosphere and soil environment control the rate at which water uptake occurs. Plants can extract the soil water in contact with the roots. Vegetables are more sensitive to water stress than other crops with high root density3.
Freshwater is used for agricultural production where it is available, but water shortage is posing a serious threat to agriculture's long-term viability, forcing it to rely more heavily on marginal water supplies, such as treated wastewater4. Wastewater irrigation has both positive and negative consequences. It can provide nutrients and organic matter to the soil and is considered an environmentally safe disposal method for treated effluents5,6. Beans require the minor elements Ca, Zn, B, Mg and Mo in addition to sufficient quantities of N, P and K7. Long-term irrigation with wastewater can alter the physical and chemical properties of the soil and the accumulation of environmental contaminants in the irrigated area8. Wastewater management can be improved to minimize the negative impacts on health and the environment9. Irrigation with wastewater contributes significantly to the accumulation of heavy metals like iron, copper, manganese and zinc in vegetables10.
Danio rerio or Zebrafish is used as a model organism in studying vertebrate genetics, developmental factors, regeneration factors and toxicological changes. The ease of care, faster breeding, short generation time, distinct developmental patterns, modes of external fertilisation and lower husbandry maintenance cost is the prime reason for using Danio rerio as model organism Danio rerio belongs to the family Cyprinidae11. Francis Hamilton, a Scottish physician, was the first to use Danio rerio in scientific experiments, describing them as "beautiful fish" with an uncertain economic value. The wild and domesticated Danio rerio have different breeding seasons. The best time to breed for wild species is from April to August, while domesticated animals will breed all year12.
Genetic studies which involve experiments on breeding and the nature and effect of mutation and clonal analysis were carried out using Danio rerio. Genetic and developmental manipulations such as androgenesis or gynogenesis are possible as they have external fertilisation. Due to the small size of adult zebrafish, evaluation of cytotoxic effects on the organism and organ system can be carried out, potentially causing the development of new metabolites13. Since zebra fish responds to chemicals in the water, it has become an important model organism for toxicology screening. Several research works on Danio rerio revealed the screening effects of potential tumour causing carcinogens. The features of the Danio rerio to encode transgenic biosensors enable it to be a more precise toxicity-screening organism14.
Histological analysis and histochemical markers extensively used to study the protein and gene expression studies in Danio rerio revealed the functional capabilities of the organ. Studies on the synthesis of intermediates such as nitric oxide or developmental neurotransmitters are carried out extensively in zebrafish15.
Morphogenesis and functional development of organ systems such as brain, eye, nose, kidney, gonad and blood vessels were analysed using Danio rerio as the model organism16. Male and female of Danio rerio exhibit distinct courtship activity that results in the release of egg and sperm at nearly the same time, resulting in a higher probability of fertilisation17. Thousands of oocytes are released, increasing fertilisation rates and turning the model organism into a prolific breeder. Zebrafish respond to chemicals in water and thus become an important model organism for toxicology screening17.
The present study was mainly focused on testing the presence and estimating the number of heavy metals in wastewater samples, to detect the toxic effect of wastewater on bean plants, to compare the toxic effects of distilled water, treated wastewater and untreated wastewater on Phaseolus vulgaris and to evaluate the toxicity of these water samples on Zebrafish fed with treated Phaseolus vulgaris.
MATERIALS AND METHODS
Study area: The study was conducted in the greenhouse and Life Science Laboratory of CHRIST (Deemed to be University) from October, 2019 to March, 2021.
Heavy metal analysis in Phaseolus vulgaris
Preparation of plant ethanolic extracts of Phaseolus vulgaris: The plants were grown in the greenhouse, divided and watered with three different water samples every 24 hrs. Distilled water, treated wastewater and untreated wastewater (Sewage treatment plant, Christ University, Bengaluru) were the water samples used in the study. It took four months for the plants to grow. Leaves and fruits of Phaseolus vulgaris were used in the study. The plant materials were washed thoroughly in running water and the leaves and fruit were air-dried followed by drying using a hot air oven at 60°C for 24 hrs. It was then powdered by a grinder and stored at room temperature. The powdered plant samples were extracted with ethanol. Concentrations of 25, 50, 100, 200 and 400 mg L–1 of leaf and fruit extracts of Phaseolus vulgaris were selected as effective concentrations for performing the main toxicity tests of the extract. The extracts were filtered using a cheese cloth and were used for further procedures18.
Analysis of heavy metals using AAS: Heavy metal analysis of water samples and powdered plant samples was done using Atomic Absorption Spectroscopy. To 100 mL of the water sample taken in a beaker, added 5 mL of concentrated HNO3. The beaker was kept at 95°C till the volume was reduced to 20 mL. The residue was filtered and made up to 100 mL. For powdered plant samples, 2 g of it was dissolved 10 mL of nitric acid, filtered the residue and made up to 100 mL. They were then loaded to the AAS spectrometer for heavy metal toxicity analysis19.
Toxicological studies on plant ethanolic extracts of Phaseolus vulgaris on Danio rerio
Oral acute toxicity of ethanolic extracts of leaves and fruits of Phaseolus vulgaris on zebrafish: The fishes were quarantined and stored in a 5 L aquarium with a water circulation system and fed with pellet feed. After 14 days of adaptation, fishes were transferred to separate test vessels with a volume of 2 L and adapted for a further 24 hrs before starting the toxicity experiments. Purified water with reverse osmosis was used in all experiments. The feeding of the fish was stopped 48 hrs before the start of the experiment to avoid the risk of absorption from the faecal material or food.
Toxicity of ethanolic leaf and fruit extracts of plant samples of Phaseolus vulgaris was tested in the zebrafish model as per the OECD guidelines 20320. The fish were exposed to the sample based on the static exposure regime. For every experiment, seven healthy fishes were directly transferred into each prepared concentration Control groups (7 fishes) were also maintained for each treatment. The mortalities were recorded at 24, 48 and 96 hrs post-exposure with different methodologies21. The dead fishes were preserved in 10% formalin.
Statistical analysis: The concentration-dependent mortality ratio was analyzed using IBM SPSS 21.0 software with 95% confidence limits. The median lethal concentration (LC50) of the acute toxicity experiment was calculated from the data using the PROBIT function and analyzed by IBM SPSS Statistics 21.0 software with 95% confidence limits. The safe level estimation after 96 hrs exposure of zebrafish to the leaf extracts of the plant was carried based on the methodology used by Hart and Sprague, Committee on Water Quality Criteria (CWQC), National Academy of Sciences/National Academy of Engineering (NAS/NAE) and International Joint Commission (IJC)22.
Histological variations on the liver of Danio rerio: After subsequent treatment with leaf and fruit extracts of Phaseolus vulgaris grown with different water samples, liver samples were dissected and stored in 10% formalin for 4-8 hrs for fixation. For processing, the dissected liver samples were placed in separately labelled cassettes in a cylinder containing 60% alcohol for 1 hr. Further processing was done using ascending grades of alcohol, 70 and 80% followed by 90% alcohol for 1 hr each. The samples were then subjected to 3 changes of absolute alcohol for 1 hr each. The samples were then exposed to 2 changes of xylol for one and half hours each. The liver tissues were placed in 2 changes of paraffin wax at 65°C for 2 hrs each. Tissue is then embedded in molten wax and allowed to solidify to make blocks. Blocks are then cut, using a microtome, to sections that are 3-5-micron thickness. Transferred paraffin ribbon carrying organ sections into a warm water bath at 45°C. Once stretched the ribbon should be scooped up onto a previously cleaned slide. After making the slide let it rest in a drying oven at 30°C. Histological slide staining with Hematoxylin and eosin (H and E staining) was done using standard protocols. When slides are ready, they can be observed under a bright-field microscope for analysis and interpretation of tissue alterations22.
RESULTS
Heavy metal analysis: Heavy metals like manganese and lead were detected in the water samples and the plant powders of Phaseolus vulgaris which were grown with those metals were used in the study using AAS Spectrophotometer. The pipe water is the treated wastewater and STP (sewage treatment plant) water is the untreated wastewater.
The concentration of Mn was found highest in STP water, i.e., 0.0457 ppm when compared to other water samples in Table 1. Mn concentration was high in the leaf and fruit powder, i.e., 0.5728 and 0.2004 ppm, respectively, of Phaseolus vulgaris treated with STP water. The concentration of Pb was found highest in STP water, i.e., 0.0191 ppm when compared to other water samples. Pb concentration was high in the leaf and fruit powder, i.e., 0.0480 and 0.0344 ppm of Phaseolus vulgaris treated with STP water in Table 2 and 3.
Toxicological studies: The study conducted on the toxicity of heavy metals revealed that the leaf and fruit extracts of Phaseolus vulgaris grown with wastewater were more toxic to Danio rerio than other extracts used in the experiment. At the observation time of 72 hrs, STP water treated leaf powder started to induce toxicity at 400 mg L–1. At the observation time of 96 hrs, pipe water treated leaf powder started to induce toxicity at 400 mg L–1. At the observation time of 96 hrs, STP water treated beans powder induced toxicity at 100 and 200 mg L–1. The LC50 of leaf extract of Phaseolus vulgaris treated with STP water was calculated as 314.598 mg L–1 at the exposure time of 96 hrs using the probit function of regression analysis in Table 4. Safe levels of the plant extracts at 96 hrs of exposure were estimated at 8.49869, 31.4598, 3.14598, 31.4598-0.00314598, 15.7299 and 15.7299 mg L–1 in Table 5.
The Pearson correlation value is 0. 959. This indicates a positive strong correlation. The significance is 0.041, which is p<0.05. This concludes that there is a correlation between concentration and mortality ratio percentage in Table 6.
Histological variations: The liver sections of Danio rerio fed with distilled water and treated wastewater grown Phaseolus vulgaris were found to be similar to the architecture of the liver of the control fish in Fig. 1 and 2.
| Table 1: Concentration of Mn in the water samples | ||
| Sample | Mn (ppm) | Pb (ppm) |
| Control (only HNO3) | 0.0018 | 0.0076 |
| Pipe water | 0.0292 | 0.0115 |
| STP water | 0.0457 | 0.0191 |
| Table 2: Concentration in leaf extracts of Phaseolus vulgaris treated with different water samples | ||
| Sample | Mn (ppm) | Pb (ppm) |
| Control (only HNO3) | 0.44 | 0.0153 |
| Pipe water | 0.5325 | 0.0573 |
| STP water | 0.5728 | 0.084 |
| Table 3: Concentration of metals in fruit extracts of Phaseolus vulgaris treated with different water samples | ||
| Sample | Mn (ppm) | Pb (ppm) |
| Control (only HNO3) | 0.1658 | 0.0038 |
| Pipe water | 0.1858 | 0.0191 |
| STP water | 0.2004 | 0.0344 |
| Table 4: Lethal concentration (LC50) of leaf extracts of Phaseolus vulgaris treated with STP water with 95% confidence limit depending on exposure time (hr) | ||||
| 95% confidence limit (mg L–1) | ||||
| Exposure (hr) | LC50 (mg L–1) | Lower | Upper | Calculated chi-square (χ2) |
| 24 | 636.086 | 812.239 | 9875.732 | 1.651 |
| 48 | 485.759 | 340.319 | 8755.787 | 1.02 |
| 72 | 402.364 | 274.682 | 1345.863 | 1.726 |
| 96 | 314.598 | 212.954 | 679.119 | 3.396 |
| Table 5: Estimation of safe levels of the plant extracts at 96 hrs of exposure | |||
| 96 hrs LC50 (mg L–1) | Method | AF | Safe level (mg L–1) |
| Hart | 8.49869 | ||
| Sprague | 0.1 | 31.4598 | |
| 314.598 | CWQC | 0.01 | 3.14598 |
| NAS/NAE | 0.1-0.00001 | 31.4598-0.00314598 | |
| CCREM | 0.05 | 15.7299 | |
| IJC | (5% of 96 hrs) LC50 | 15.7299 | |
*C: 48 hrs LC50×0.03/S2 where, C is the presumable harmless concentration and S = 24 hrs LC50/48 hrs LC50 | |||
| Table 6: Pearson correlation between concentration and mortality ratio percentage | |||
Concentration | Mortality (%) | ||
| Concentration | Pearson correlation | 1 | 0.959* |
| Sig. (2-tailed) | 0.041 | ||
| N | 4 | 4 | |
*Correlation is significant at the 0.05 level (2-tailed) | |||
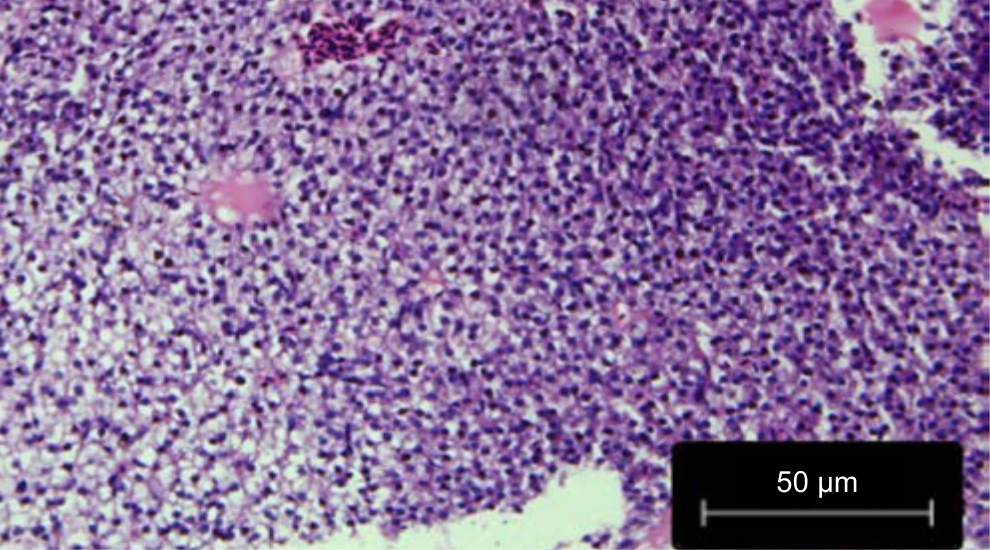 |
| Fig. 1: | Control: Liver treated with leaf extracts of Phaseolus vulgaris treated with distilled water |
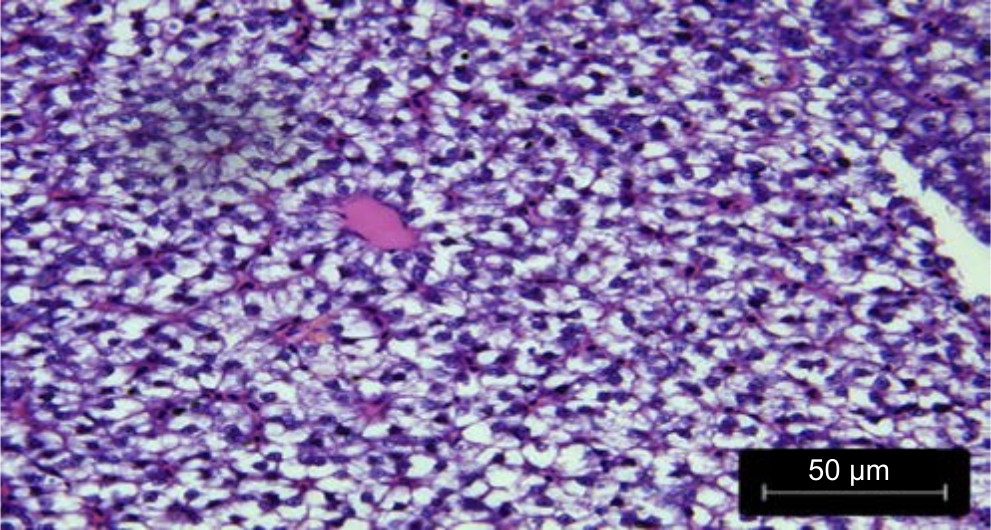 |
| Fig. 2: | Control: Liver treated with fruit extracts of Phaseolus vulgaris treated with distilled water |
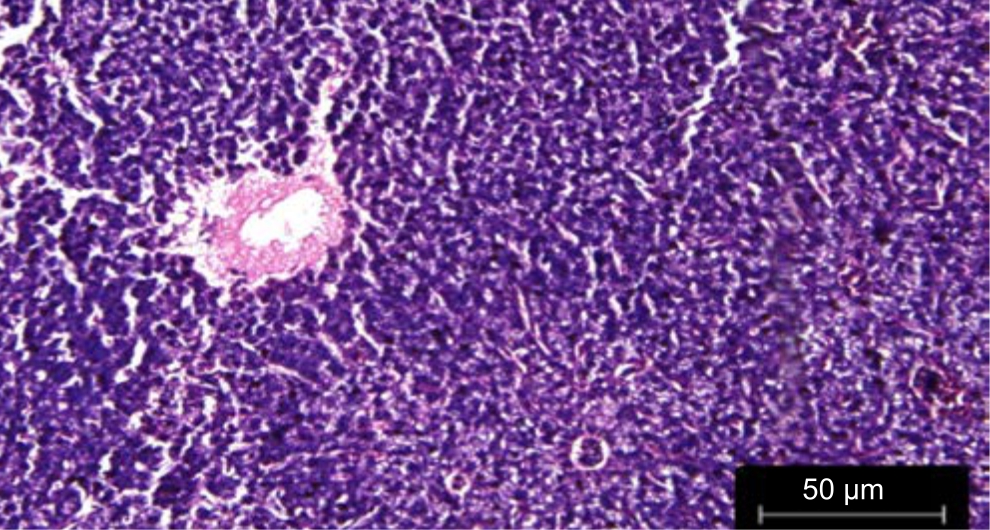 |
| Fig. 3: | Liver treated with leaf extracts of Phaseolus vulgaris irrigated with wastewater |
The liver of Danio rerio fed with leaf extracts of Phaseolus vulgaris irrigated with wastewater did not show much variation or histopathological variation and was found to be similar to the control in Fig. 3. There was no histopathological variation l in the liver sections treated with fruit extracts of Phaseolus vulgaris irrigated with treated wastewater in Fig. 4.
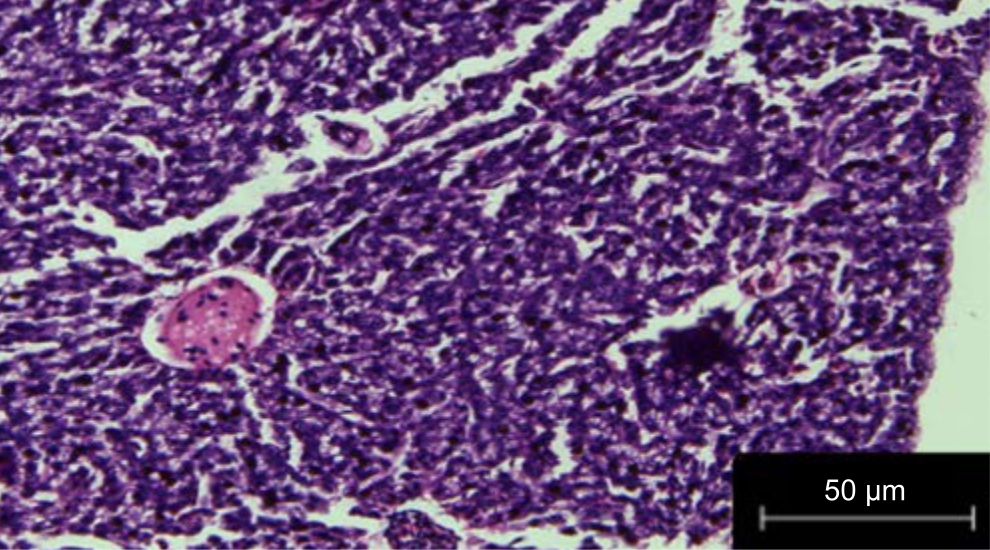 |
| Fig. 4: | Liver treated with fruit extracts of Phaseolus vulgaris irrigated with treated wastewater |
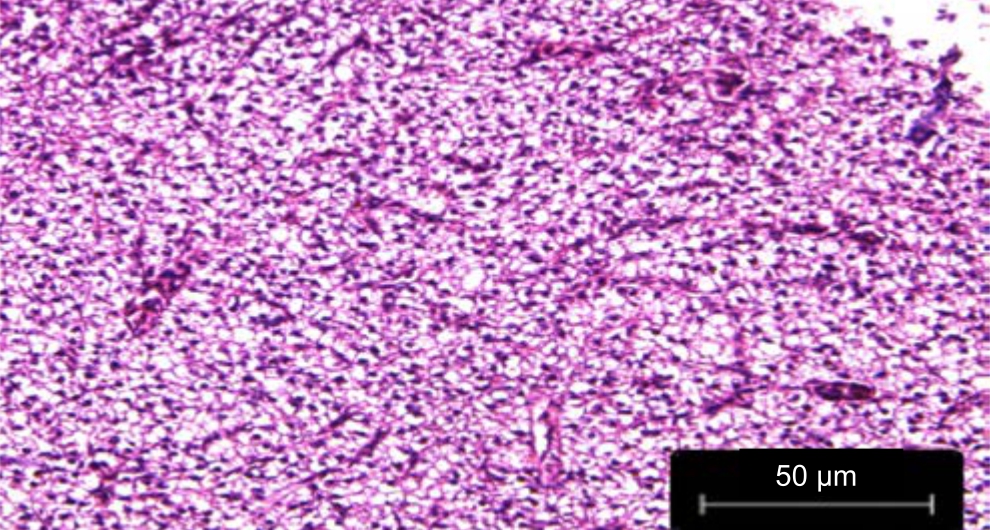 |
| Fig. 5: | Liver treated with leaf extracts of Phaseolus vulgaris irrigated with untreated water |
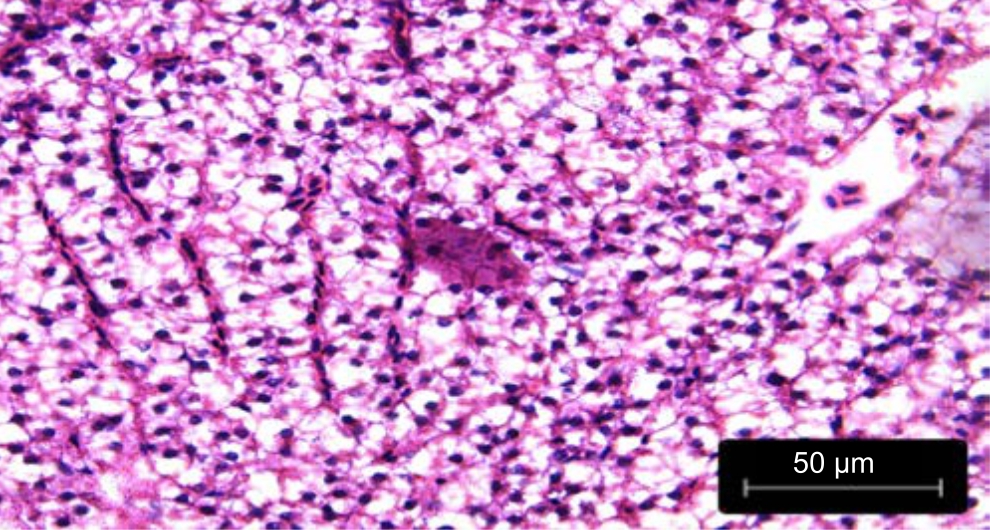 |
| Fig. 6: | Liver treated with fruit extracts of Phaseolus vulgaris irrigated with untreated water (40X) |
The congestion of the central vein and loss of the cord arrangement of hepatocytes around blood capillaries were observed in the liver sections of the Danio rerio fed with leaf extracts of Phaseolus vulgaris irrigated with untreated wastewater in Fig. 5. Cell clearing and striations were also observed along with congestion of the central vein in Danio rerio fed with the fruit extracts of Phaseolus vulgaris irrigated with untreated wastewater in Fig. 6.
DISCUSSION
The present study was focused mainly on three facts concerning the three water samples. The attempt was made to estimate the number of heavy metals present in wastewater samples, to compare the toxic effects of distilled water, treated wastewater and untreated wastewater on Phaseolus vulgaris and to evaluate the toxicity of these water samples on Zebrafish. Heavy metals like manganese and lead were detected and estimated using AAS in water samples and the plant's parts irrigated with it.
Plant availability of manganese depends on soil adsorption and Mn reduction. Soils with higher manganese sorption capacity have a lower potential for plant absorption of manganese23.
Plants absorb and accumulate lead in roots, stems, leaves, root nodules and seeds with an increase in its exogenous level. The majority of lead absorbed by plants reside in roots and only a small fraction of it is translocated to the shoots24.
Soil with low pH (3.9) caused increased solubility of lead that resulted in its higher uptake25. A high concentration of lead in soils can affect soil productivity. The plant processes like photosynthesis, mitosis and water absorption are affected at even very low lead concentrations showing toxic symptoms like dark leaves, wilting of older leaves, stunted foliage and brown short roots26.
Long term exposure to lead resulted in reduced leaf growth, decreased photosynthetic pigments, changed chloroplast structure and decreased enzyme activities required for CO2 accumulation. Transpiration intensity, the osmotic pressure of cell sap, the water potential of xylem and relative water content are the other factors that were significantly reduced27.
Heavy metals, such as arsenic, cadmium, lead and mercury, are the most harmful to humans, wildlife, fish and the environment. Heavy metal concentrations that are too high are harmful. Because of their bioaccumulation in animals, adverse effects on biota and even mortality of most living creatures, they destabilise habitats28.
Toxic heavy metals enter the human body by the consumption of plants containing accumulated levels of heavy metals29. Heavy metal contaminated food can seriously deplete some of the essential nutrients in the body that can finally lead to decreasing immunological defences, intrauterine growth retardation, impaired psychosocial faculties, disabilities associated with malnutrition and high prevalence of upper gastrointestinal tract30.
The sensitivity of leaf and fruit extracts of Phaseolus vulgaris on experimental fish have shown that the lethal effect of a leaf extract from plants treated with wastewater was highest on Zebrafish at a concentration of 400 mg L–1. The present study revealed the fact that the leaf and fruit of Phaseolus vulgaris grown with distilled water is safe to consume at even higher concentrations. The leaves of Phaseolus vulgaris irrigated with treated wastewater are safe to consume in a limited dose, that is, up to 400 mg L–1 in Danio rerio.
The leaves demonstrated greater toxicity than fruits of the plant, which proves that heavy metals accumulate more in leaves than fruits or other parts of the plant.
Heavy metals are significant contaminants for fish because they are not removed from water environments using natural processes, such as biological pollutants, which are enriched in mineral organic compounds. Heavy metals are found in different amounts in fish based on their age, growth and other physiological factors. Fish are one of the animal species that can be severely harmed by these chemical contaminants. Heavy metals can be poisonous to a variety of organs. Heavy metals join the biogeochemical cycle as they become more concentrated in the atmosphere, causing toxicity. Behavioural tests revealed several signs linked to such elemental anomalies, such as lack of appetite, constipation, salivation, photophobia and a proclivity to walk in a circle, among others. Mutagenicity, carcinogenicity, teratogenicity, immunosuppression, poor body condition and compromised fertility are the most common toxic effects associated with chronic exposure to pollutant heavy metals31.
Elements like copper, titanium, cadmium and zinc, have significant harmful effects on humans and animals, in addition to lead. Metal toxicity has been studied extensively using Zebrafish and a unique transgenic animal has been created to detect the presence of metals in water14.
The accumulation of heavy metals like Fe, Mn, Zn, Cu, Pb and Cd on liver tissues showed dilation and congestion in blood vessels and blood sinusoids, haemorrhages in hepatocytes, hemolysis and hemosiderin pigment, degeneration of necrosis in hepatocytes, fatty degeneration in hepatocytes and parasite cysts in the liver tissues of Tilapia zilli. Liver cells damage also lead to increased GOT and GPT enzyme activities associated with it31.
The liver cells of Danio rerio have homogenous cytoplasm with a large spherical nucleus and blood capillaries called sinusoids. The photomicrographs of Zebrafish treated with metals showed necrosis, degenerative nuclei and cytoplasmic vacuolation32.
The existence of eosinophilic granules in the cytoplasm, irregular-shaped nuclei, nuclear hypertrophy, nuclear vacuolation and eosinophilic granules in the nucleus were the most common changes observed in the liver. Cytoplasmic and nuclear degeneration was also very common32.
The histopathological sections of the liver of rats showed the degeneration of hepatocytes, congestion of the central vein and changes in the cord arrangement. In rats treated with high doses of lead acetate, hepatocytes focal necrosis, fatty degeneration and portal tract with massive aggregation of mononuclear inflammatory cells scattered in different spaces were observed. The photomicrographs of liver sections of rats stained with H and E treated with a low dose of aluminium chloride showed moderate hydropic degeneration of hepatocytes and those treated with high doses of aluminium chloride showed fragmented hepatocytes necrosis with nuclei that finally lead to necrotic hepatocytes infiltrated with inflammatory cell32.
CONCLUSION
The present study revealed that the leaf and fruit extracts of Phaseolus vulgaris grown with untreated wastewater were more toxic to Danio rerio than other extracts used in the experiment. In experiments carried out with leaf and fruit extracts of Phaseolus vulgaris grown with distilled water, no mortality was observed and was found to be safe to consume. The leaf extracts of plants grown with treated wastewater showed mortality at 400 mg L–1. It can be concluded that the leaf extracts of Phaseolus vulgaris grown with both treated and untreated wastewater is toxic to fishes and thus can be toxic to humans at higher concentrations. More studies should be performed to prove more scientifically the toxicity level of wastewater irrigated leaves and fruits of Phaseolus vulgaris in human beings.
SIGNIFICANCE STATEMENT
“This study discovered the presence of toxicity in Phaseolus vulgaris irrigated with wastewater. This study will help the researchers to uncover the critical areas of toxicity levels in vegetables that are irrigated with wastewater in underdeveloped and developing countries”.
REFERENCES
- Argaw, A. and D. Muleta, 2018. Effect of genotypes-Rhizobium-environment interaction on nodulation and productivity of common bean (Phaseolus vulgaris L.) in eastern Ethiopia. Environ. Syst. Res., Vol. 6.
CrossRefDirect Link - Wang, L., Y. Cao, E.T. Wang, Y.J. Qiao and S. Jiao et al., 2016. Biodiversity and biogeography of rhizobia associated with common bean (Phaseolus vulgaris L.) in Shaanxi Province. Syst. Appl. Microbiol., 39: 211-219.
CrossRefDirect Link - Barabaschi, D., A. Tondelli, F. Desiderio, A. Volante, P. Vaccino, G. Valè and L. Cattivelli, 2016. Next generation breeding. Plant Sci., 242: 3-13.
CrossRefDirect Link - Allende, A. and J. Monaghan, 2015. Irrigation water quality for leafy crops: A perspective of risks and potential solutions. Int. J. Environ. Res. Public Health, 12: 7457-7477.
CrossRefDirect Link - Xu, J., L. Wu, A.C. Chang and Y. Zhang, 2010. Impact of long-term reclaimed wastewater irrigation on agricultural soils: A preliminary assessment. J. Hazard. Mater., 183: 780-786.
CrossRefDirect Link - Horswell, J., T.W. Speir and A.P. van Schaik, 2003. Bio-indicators to assess impacts of heavy metals in the land-applied sewage sludge. Soil Biol. Biochem, 35: 1501-1505.
CrossRef - Balestre, M., P.P. Torga, R.G.V. Pinho and J.B. dos Santos, 2012. Applications of multi-trait selection in common bean using real and simulated experiments. Euphytica, 189: 225-238.
CrossRefDirect Link - Rattan, R.K., S.P. Datta, P.K. Chhonkar, K. Suribabu and A.K. Singh, 2005. Long-term impact of irrigation with sewage effluents on heavy metal content in soils, crops and groundwater: A case study. Agric. Ecosyst. Environ., 109: 310-322.
CrossRefDirect Link - Qadir, M., D. Wichelns, L. Raschid-Sally, P.G. McCornick, P. Drechsel, A. Bahri and P.S. Minhas, 2010. The challenges of wastewater irrigation in developing countries. Agric. Water Manage., 97: 561-568.
CrossRefDirect Link - Arora, M., B. Kiran, S. Rani, A. Rani, B. Kaur and N. Mittal, 2008. Heavy metal accumulation in vegetables irrigated with water from different sources. Food Chem., 111: 811-815.
CrossRefDirect Link - Zhang, C., C. Willett and T. Fremgen, 2003. Zebrafish: An animal model for toxicological studies. Curr. Protoc. Toxicol., Vol. 17.
CrossRefDirect Link - Stevens, C.H., B.T. Reed and P. Hawkins, 2021. Enrichment for laboratory Zebrafish—a review of the evidence and the challenges. Animals, Vol. 11.
CrossRefDirect Link - Spitsbergen, J.M. and M.L. Kent, 2003. The state of the art of the zebrafish model for toxicology and toxicologic pathology research—advantages and current limitations. Toxicol. Pathol., 31: 62-87.
CrossRefDirect Link - Bambino, K. and J. Chu, 2017. Zebrafish in Toxicology and Environmental Health. In: Current Topics in Developmental Biology, Sokol, S.Y. (Ed.)., Elsevier Inc., pp: 331-367.
CrossRefDirect Link - Cole, L.K. and L.S. Ross, 2001. Apoptosis in the developing zebrafish embryo. Dev. Biol., 240: 123-142.
CrossRefDirect Link - Osborne, N., G. Paull, A. Grierson, K. Dunford and E.M. Busch-Nentwich et al., 2016. Report of a meeting on contemporary topics in zebrafish husbandry and care. Zebrafish, 13: 584-589.
CrossRefDirect Link - Darrow, K.O. and W.A. Harris, 2004. Characterization and development of courtship in zebrafish, Danio rerio. Zebrafish, 1: 40-45.
CrossRefDirect Link - Sukender, K., S. Jaspreet, D. Sneha, and G. Munish, 2012. AAS estimation of heavy metals and trace elements in Indian herbal cosmetic preparations. Res. J. Chem. Sci., 2: 46-51.
Direct Link - LaFollette, M.R., M.C. Riley, S. Cloutier, C.M. Brady, M.E. O'Haire and B.N. Gaskill, 2020. Laboratory animal welfare meets human welfare: A cross-sectional study of professional quality of life, including compassion fatigue in laboratory animal personnel. Front. Vet. Sci., Vol. 7.
CrossRefDirect Link - Avdesh, A., M. Chen, M.T. Martin-Iverson, A. Mondal and D. Ong et al., 2012. Regular care and maintenance of a zebrafish (Danio rerio) laboratory: An introduction. J. Vis. Exp., Vol. 69.
CrossRefDirect Link - Xavier, J. and K. Kripasana, 2020. Acute toxicity of leaf extracts of Enydra fluctuans Lour in Zebrafish (Danio rerio Hamilton). Scientifica, Vol. 2020.
CrossRefDirect Link - Patra, M., N. Bhowmik, B. Bandopadhyay and A. Sharma, 2004. Comparison of mercury, lead and arsenic with respect to genotoxic effects on plant systems and the development of genetic tolerance. Environ. Exp. Bot., 52: 199-223.
CrossRefDirect Link - Ernst, W.H.O., H.J.M. Nelissen and W.M.T. Bookum, 2000. Combination toxicology of metal-enriched soils: Physiological responses of a Zn- and Cd-resistant ecotype of silene vulgaris on polymetallic soils. Environ. Exp. Bot., 43: 55-71.
CrossRefDirect Link - Kanwal, A., M. Farhan, F. Sharif, M.U. Hayyat, L. Shahzad and G.Z. Ghafoor, 2020. Effect of industrial wastewater on wheat germination, growth, yield, nutrients and bioaccumulation of lead. Sci. Rep., Vol. 10.
CrossRefDirect Link - Govind, P. and S. Madhuri, 2014. Heavy metals causing toxicity in animals and fishes. Res. J. Anim. Vet. Fishery Sci., 2: 17-23.
Direct Link - Ghosh, A.K., M.A. Bhatt and H.P. Agrawal, 2012. Effect of long-term application of treated sewage water on heavy metal accumulation in vegetables grown in Northern India. Environ. Monit. Assess., 184: 1025-1036.
CrossRefDirect Link - Iyengar, G.V. and P.P. Nair, 2000. Global outlook on nutrition and the environment: Meeting the challenges of the next millennium. Sci. Total Environ., 249: 331-346.
CrossRefDirect Link - Ibrahim, S. and S. Mahmoud, 2005. Effect of heavy metals accumulation on enzyme activity and histology in liver of some nile fish in Egypt. Egypt. J. Aquat. Biol. Fish., 9: 203-219.
CrossRefDirect Link - Bhuvaneshwari, R., K. Padmanaban and R.B. Rajendran, 2015. Histopathological alterations in muscle, liver and gill tissues of zebra fish danio rerio due to environmentally relevant concentrations of organochlo rine pesticides (OCPs) and heavy metals. Int. J. Environ. Res., 9: 1365-1372.
CrossRefDirect Link - Camargo, M.M.P. and C.B.R. Martinez, 2007. Histopathology of gills, kidney and liver of a Neotropical fish caged in an urban stream. Neotrop. Ichthyol., 5: 327-336.
CrossRefDirect Link - Goma, A.A.E.M. and H.G. Tohamy, 2016. Impact of some heavy metals toxicity on behaviour, biochemical and histopathological alterations in adult rats. Adv. Anim. Vet. Sci., 4: 494-505.
CrossRefDirect Link








