Research Article
Hepatoprotective Effects of Royal Jelly against Hepatotoxicity and Oxidative Stress Induced by Sodium Fluoride in Male Rats
Department of Biological Sciences, Zoology, King Abdul-Aziz University, Jeddah 21589, Saudi Arabia
Fluorine is not present-free. Fluorine (F) in drinking water is found in ionic form and hence it passively passes through the intestine and interferes with metabolic pathways of the living system. The F in small doses has remarkable prophylactic influence, while in higher doses, it causes skeletal fluorosis1.
The F enters the body via drinking water, toothpaste and food, fluoride dust and fumes from industries by using fluoride-containing salt. People are currently at risk worldwide because they drink water containing amounts of F2. The permissible limit of F is 1 mg L–1 according to WHO guidelines3. Exposure to F results in the generation of anion superoxide (O2–), elevated O2 concentration and its downstream consequences such as H2O2. Although several hypotheses have been proposed, the exact mechanism of F toxicity has not been clearly defined.
Experimental evidence has indicated that exposure to F results in oxidative stress in tissues such as the liver, brain and testes. Fluorine inhibits the activities of antioxidant enzymes SOD, GPx and CAT and GRx4. The high toxicity of sodium fluoride arises from its being a very reactive ion. The F crosses the cell membrane very rapidly5 and is distributed in the skeletal and cardiac muscle, liver and erythrocytes6,7. A study indicated that stress and inhibition of protein synthesis produced by F during dental enamel development were the main cause of dental fluorosis in the mice incisors that were given water containing 50-100 ppm of F daily for successive 30 days8.
Supplementation of sodium fluoride (50 mg L–1) to drinking water for 6 months produced severe oxidative stress and apoptosis in male rats8. This may contribute to an elevation of the level of F in the serum of animals that received sodium fluoride. The F’s chronic toxic action has also been investigated in the hepatic tissues. Oxidative stress was produced by NaF in the liver of rabbits8.
Fluoride was determined to cause adverse effects in mice on erythrocyte and liver tissue9. They also reported that lipid biomarkers and AST, ALT and ALP activities were affected.
In NaF-treated male rats (10 mg/kg/day, for successive 30-50 days), declined sperm motility and a reduction in serum testosterone levels have been reported10. Hsiao et al.11 noticed that F may be a toxicant in many body tissues. The use of natural antioxidant phytochemicals has proven to be an effective and safe dietary reference for hepatic disease12. A major phytochemical class is flavonoids, which are highly efficient against reactive oxygen species-induced injury13. Thus, flavonoids are considered for the prevention and treatment of hepatic diseases.
The RJ is a fluid released by the mandibular and hypopharyngeal glands of worker bees. The worker bee larvae then begin to ingest worker jelly, which is largely consisting of honey and pollen14. That is the food that all honeybee larvae eat for the 1st 3 days after they are born.
The RJ is a mixture of multi-components such as minerals, glucose, lipids, vitamins, nucleic acids and acetylcholine15, which are essential for the properties of RJ16.
The RJ is essentially utilized to save human health17. Additionally, jelly is a type of functional food and dietary supplement with various biological actions, including anti-allergy and immunological modulation. These functions are linked to RJ’s many functional ingredients, including proteins, lipids, carbs, vitamins and minerals18. The RJ has significant multi-protective benefits in male Wister albino rats intoxicated with different toxicants by avoiding oxidative injury and improving the former biochemical parameters19.
Because of its critical involvement in the metabolism and clearance of foreign chemicals, the liver plays a critical role in determining drug toxicity. Cytochrome P450 enzymes (CYP450) metabolize most of the xenobiotic compounds in the liver20. However, many parameters can impact the hepatic tissue’s metabolic functions and induction is a common cause of drug-drug interactions21. So, the present study objective was to investigate the effect of the RJ on the hepatotoxicity induced by NaF or their combination in male rats using chemico-biological markers of liver injury, serum lipids, oxidative stress, inflammation and histological structures.
Study area: This study was carried out at the Department of Biological Sciences, Zoology, King Abdul-Aziz University, Saudi Arabia in 2021/2022.
Chemicals: Pure royal jelly capsules were purchased from Pharmaceuticals Co., Saudi Arabia. Whereas, sodium fluoride (NaF) was provided by Sigma-Aldrich, St. Louis, MO, USA.
Animals groups, ethics and treatment: The animals were cared for following the ethics guidelines for laboratory animal care (approval No. 42-0073). A total of 40 male rats weighing between 150 and 170 g were obtained and kept at the animal house. The rats were placed under typical laboratory conditions (22°C, 50% relative humidity and a natural daylight cycle of 12/12 hrs). They were fed with a commercially available pellet diet. Food and drink were freely accessible.
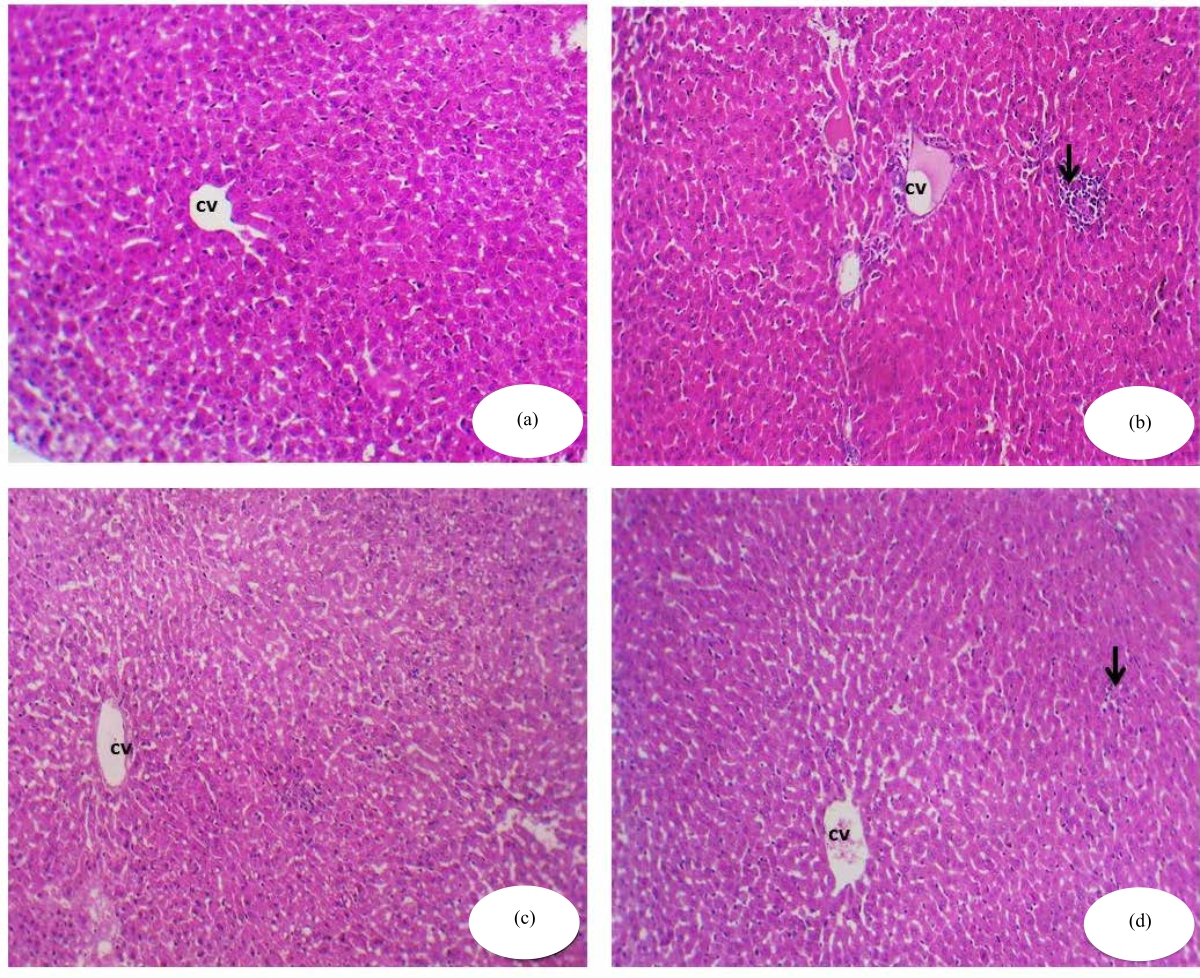 |
| Fig. 1(a-d): | Histopathological view of the rat liver in different groups, (a) Control group: Normal hepatic architecture, central vein (cv) and cords of hepatocytes (H&E ×200), (b) Photomicrograph of a cross-section of experimental rat liver treated with NaF showing lobular inflammation ( arrow) and hepatocytic apoptosis (H&E ×200), (c) Photomicrograph of a cross-section of the hepatic tissues of RJ treated group showing normal hepatic structure (H&E ×200) and (d) Photomicrograph of a cross-section of hepatic tissues of experimental rat liver treated with the combination of NaF and RJ showing an almost normal central vein lined by endothelial cells and hepatic cords radiating from the central vein, showing hepatocytic apoptosis (H&E x200) |
The experiment was performed on 40 male albino rats, divided into four groups and was all administrated the compounds orally (by using a rat oral feeding tube) for successive 30 days (Scheme 1 and Fig. 1):
| • | Group I served as the control, the animals received 0.9% NaCl (physiological saline) |
| • | Group II was given 10.3 mg kg–1 of NaF 4 dissolved in physiological saline |
| • | Group III was given royal jelly (RJ, 85 mg kg–1 diluted in saline and this dose is equivalent to 250 mg crude RJ)22 |
| • | Group IV received NaF as the first dosage, followed by RJ 30 min after the NaF was administered. All animals were treated orally for 30 successive days |
Sample collection: To separate the serum, blood samples were taken and centrifuged at 3000 rpm for 10 min at 4°C. The obtained serum was kept at 20°C for further testing. The rats were quickly decapitated after mild anaesthesia with xylene/ketamine HCl (75 and 10 mg kg–1 intraperitoneally) and the liver was homogenized. After centrifuging homogenates at 4500 g for 20 min at 4°C, the supernatants were collected and kept at 20°C for future antioxidant enzyme investigation.
Biochemical investigation
Liver function assessment and inflammation markers: On the 30th day of treatment, serum Lactate Dehydrogenase (LDH) activity of 6 hrs post-fasted rats was assessed. Serum LDH activity was determined using a spectrum diagnostics LDH reagent/assay kit (MDSS GmbH Schiffgraben, Hannover, Germany) according to the manufacturer’s instructions. Also, the activity of Alanine Aminotransferase (ALT), Aspartate Aminotransferase (AST) and Alkaline Phosphatase (ALP) were evaluated in serum using commercial kits (Spinreact, Spain) according to the manufacturer’s instructions.
The lipid profile was evaluated by the determination of serum total cholesterol (TC) and triglycerides (TG) according to Farag et al.23 and Alagawany et al.24. Low-density lipoprotein cholesterol (LDL-c) and Very-low-density Lipoprotein Cholesterol (vLDL-c) were determined25,26.
The sandwich Enzyme-Linked Immunosorbent Assay (ELISA) technique was conducted according to the manufacturer’s (Ebio-Science) instructions (Bio-Tek Instruments, Inc.) to determine interleukin 6 (IL-6) (Cat. No. BMS625), tumour necrosis factor-alpha (TNF-α) (Cat. No. BMS622) and C-reactive protein (CRP).
Assessment of antioxidant and oxidative stress indices: A small piece of the liver (0.30 g) was separated and homogenized using cold Tris-HCl buffer (pH 7.4). It was centrifuged to obtain the supernatant used for further biochemical assays. According to Farag et al.27, malondialdehyde (MDA) was detected as a lipid peroxidation (LPO) product using thiobarbituric acid reactive compounds.
According to Sun et al.28, superoxide dismutase (SOD), activity was assessed. The degree of inhibition of this process is then used to calculate SOD activity. Aebi29 published the technique for determining CAT activity, which involves measuring the rate of breakdown of H2O2 at 240 nm (Spectrophotometer SP-2200, Bioespectro). The results were reported in U g–1 protein. Glutathione peroxidase (GPx) activity was assessed.
Histological assessment: Fixed samples of hepatic tissues were processed following a conventional procedure for hematoxylin/eosin staining of liver tissue sections. A light microscope was used to view the stained slides and photomicrographs of the tissue samples were taken.
Statistical analysis: The data were expressed as Mean±SE. For data comparison comparing several groups, a One-way Analysis of Variance was employed, using a least significant difference post hoc test. Statistical significance was defined as a value of p<0.05.
The present study was conducted to evaluate the adverse effects of NaF alone or in combination with RJ on liver biomarkers. The antioxidant enzymes such as SOD, CAT and GPx and the liver tissues, were used to examine histopathological and biochemical variations.
Biochemical evaluation: Table 1 shows the serum enzyme activity results of all groups. The AST, ALT, ALP and LDH activities were increased in the NaF treated group than in the control group. Treatments with RJ elevated the protein content in the NaF group. The total protein level decreased in NaF-treated rats compared to the control group. The 85 mg kg–1 of RJ was determined to be nontoxic for the enzymatic activity. A decline in the enzymatic activity was observed when the rats were treated with RJ after NaF administration.
All the lipid profiles increased with the effect of NaF (Table 2). There was a significant decrease in serum TG in groups treated with NaF and RJ compared to NaF alone. The administration of NaF resulted in liver dysfunction caused by increasing the levels of TG, TC, LDL-c and vLDL-c. On the other hand, HDL-c levels declined and enzymes’ leakage from hepatocytes increased, indicative of cellular damage. The group treated with NaF and RJ could restore all the previous parameters. In addition, RJ protected the liver cells in the group treated with NaF.
The CRP, IL-6 and TNF-α levels were significantly higher in the NaF group concerning the control animal (Table 3). The levels of CRP, IL-6 and TNF-α were significantly lower in all RJ groups compared to the high-inflammatory groups treated with NaF (Table 3), causing liver damage. As a result of an increase in pro-inflammatory markers, they may have a role in the pathogenesis of various liver illnesses. In the current study, CRP, TNF-α and IL-6 levels were notably higher in NaF-treated rats than in the control.
Table 4 shows the hepatic malondialdehyde (MDA) level as well as catalase (CAT), superoxide dismutase (SOD) and glutathione peroxidase (GPx) activities. The MoO3-NPs group had considerably greater MDA levels in liver tissue than the control group but significantly reduced CAT, SOD and GPx activities. These results indicated that MDA levels in the hepatic tissues were markedly elevated and antioxidant enzyme levels were markedly declined in response to treatment with NaF. Results also showed that NaF induced oxidative stress in the hepatic tissues of male rats, which is an indicator of an increment in the level of lipid peroxidation (LPO) and MDA level and a decrement in the antioxidant enzyme levels of CAT or GPx and/or SOD.
| Table 1: | Assessment of RJ on serum enzyme activity of male rats treated with NaF or their combinations for 30 successive days | |||
| Parameters | Control | NaF | RJ | NaF+RJ |
| ALT (U L–1) | 13.12±0.87c | 194.80±9.05a | 13.15±0.74c | 20.14±2.22b |
| AST (U L–1) | 12.45±0.76c | 293.50±8.86a | 12.15±0.62c | 20.30±2.83b |
| ALP (U L–1) | 23.27±0.94c | 159.72±7.07a | 23.07±3.70c | 28.91±3.85b |
| LDH (U L–1) | 102.04±8.29c | 654.17±26.12a | 100.43±10.51d | 111.60±8.91bc |
| Total proteins (g dL–1) | 7.85±1.55ab | 3.34±0.91d | 7.77±1.30b | 7.46±1.17c |
| Values are expressed as Mean±SE, n = 10 for each treatment group, NaF: Sodium fluoride, RJ: Royal jelly, ALT: Alanine aminotransferase, AST: Aspartate aminotransferase, ALP: Alkaline phosphatase, LDH: Lactate dehydrogenase and symbols of different alphabetical are indicated to be significantly different as compared to the control group and other treated groups (p<0.05) | ||||
| Table 2: | Effect of RJ on the serum lipid profile of male rats given NaF or a combination for 30 successive days | |||
| Parameters (mg dL–1) | Control | NaF | RJ | NaF+ RJ |
| TG | 76.01±1.66b | 184.11±10.56a | 73.17±2.04b | 110.19±8.23c |
| TC | 134.19±4.40b | 264.16±7.99a | 122.21±9.25c | 139.76±5.12b |
| HDL-c | 39.30±0.60a | 25.78±2.866d | 36.88±1.41b | 34.11±4.19c |
| LDL-c | 28.91±1.02b | 38.94±3.84a | 27.19±1.36c | 27.75±1.01c |
| vLDL-c | 15.41±0.211c | 38.34±2.60a | 14.33±1.44c | 22.14±0.47b |
| Values are expressed as Mean±SE, n = 10 for each treatment group, NaF: Sodium fluoride, RJ: Royal jelly, HDL-c: High-density lipoprotein, LDL-c: Low-density lipoprotein, vLDL-c: Very-low-density lipoprotein and symbols are different alphabetically to indicate a significant comparison to the control group and other treated groups (p<0.05) | ||||
| Table 3: | Effects of royal jelly on pro-inflammatory serum levels in male rats treated with NaF or combinations of the two for 30 successive days | ||
| Groups | IL-6 (pg g–1) | TNF-α (pg g–1) | CRP (mg L–1) |
| Control | 3.30±0.13c | 5.28±0.10c | 4.24±0.11c |
| NaF | 22.98±0.59a | 36.79±0.69a | 28.81±0.49a |
| RJ | 3.02±0.03c | 5.74±0.28c | 4.74±0.27c |
| NaF+RJ | 7.33±0.53b | 15.85±0.399b | 13.81±0.91b |
| Values are expressed as Mean±SE, n = 10 for each treatment group, NaF: Sodium fluoride, RJ: Royal jelly, TNF-α: Tumor necrosis factor alpha, IL-6: Interleukin-6, CRP: C-reactive protein and symbols are arranged alphabetically to indicate significant comparisons compared to the control group and other treated groups (p<0.05) | |||
| Table 4: | Effect of RJ on the oxidant and antioxidant status of male rats’ livers after treatment with NaF and combinations of the two for 30 successive days | |||
| Groups | MDA (nmoles of MDA g–1) | CAT (nmol g–1 of protein min–1) | SOD (U g–1 of protein) | GPx (nmol g–1 of protein min–1) |
| Control | 3.43±0.82c | 7.19±1.27bc | 18.19±1.72b | 13.11±1.29a |
| NaF | 40.53±3.68a | 3.25±0.74d | 6.40±1.19d | 5.36±1.08c |
| RJ | 3.60±0.75c | 8.07±0.81a | 20.66±1.65a | 14.15±1.39a |
| NaF+RJ | 10.13±1.96b | 7.10±0.45c | 16.10±1.58c | 12.45±0.78b |
| Values are expressed as Mean±SE, n = 10 for each treatment group, NaF: Sodium fluoride, RJ: Royal jelly, SOD: Superoxide dismutase, MDA: Malondialdehyde, GPx: Glutathione peroxidase, CAT: Catalase and symbols are different alphabetically to indicate a significant comparison to the control group and other treated groups (p<0.05) | ||||
Histopathological alterations: After administration of NaF and RJ, rat liver showed toxicity in the form of fatty change, hydropic degeneration in some hepatocytes, ductular reaction at the periphery of the portal tract and infiltration of blood sinusoids by mononuclear inflammatory cells. After administration of NaF and RJ, the liver showed moderate toxicity in the form of hypertrophy of hepatocytes with the appearance of binucleated hepatocytes and increased eosinophilia, focal necrosis in some hepatocytes and the central vein showed marked dilatation filled with haemorrhage. The rats treated with the combination of NaF followed by RJ showed an almost normal central vein lined by endothelial cells and hepatic cords radiating from the central vein. Blood sinusoids, bordered by flat endothelial cells, separate the cords (Fig. 1a-d).
The present study was an attempt to evaluate the toxic effect of NaF on liver tissues in male rats and the possible ameliorative role of RJ as it is well known that RJ has been reported to be an effective antioxidant, therefore, the present study aimed to elucidate the possible ameliorative role of RJ in alleviating the toxicity of NaF when given to normal rats.
The liver biomarkers were detected in the present study using some enzyme activity and the ability to synthesize the protein. The increase in AST, ALT and ALP activities was related to hepatic damage and the change in hepatic functions. ALP activity elevates in case of the damage to hepatic cells4. Liver cells contain more AST than ALT, but ALT is confined to the cytoplasm, in which its concentration is higher than that of AST. F toxicities caused elevated transaminases’ activities in rats and mice30,31.
Several previous studies confirmed the concept of the beneficial and antioxidant effects of RJ32-34. Therefore, the present study was carried out to study the effects of NaF and their impact on hepatic functions, histological structures and antioxidant enzyme levels. The current findings are of significant importance to alleviate the severe oxidative stress series which is the cause of risky diseases due to exposure to any toxins.
The hepatic functions enzymes (AST, ALT, ALP and LDH) indicated that cell membrane disruption occurred35,36 after treatment with NaF. The decline in total protein also might be caused by inflammation of the hepatic tissues due to NaF exposure, which greatly disturbs protein biosynthesis37.
The ALT is an enzymatic marker of hepatic damage38. In the current study, the administration of NaF led to a significant increment in ALT levels. Glucoprotein is a glycoprotein found in RJ, it plays a vital role in stimulating liver regeneration and hepatocyte improvement. Previous studies demonstrated that RJ has a strong hepatoprotective effect against xenobiotics, leading to hepatic damage39,40. The hepatoprotective effects of RJ observed in this study are in complete agreement with previous results reported by Mahesh et al.38, who demonstrated that RJ declined oxidative injury in the hepatic tissues through an imbalance in the oxidant status, causing oxidation of biological biomolecules. The NaF exposure induces acute hepatotoxicity, apoptosis, necrosis and hepatic infiltration, as shown in histological and histochemical sections41.
Recently, the function of MRJPs of RJ, antimicrobial properties42, medicinal value and health ageing43 have been reported and t. The MRJP is a weak acidic glycoprotein that accounts for approximately 48% of RJ proteins. Thus RJ is considered a protective agent for heart vitality and adjusting lipid levels in the body. Dyslipidemia is a greater factor for heart disease aggravated by a poor diet. Atherosclerotic cardiovascular disease is caused by low HDL-c and high TG and LDL-c levels in the blood44. Many types of research have been conducted to see how RJ affects blood lipid concentrations and they have discovered that RJ can lower TC levels while increasing HDL-c levels45-47. Free radicals cause the development of many diseases with harmful effects that cause cellular component peroxidation, which is tissue destruction. Antioxidants may prevent several diseases caused by free radicals48.
The RJ as an antioxidant, can inhibit lipid peroxidation that facilitates the progression of a lot of diseases49. Previous studies showed that RJ proteins have antioxidant activity against lipid peroxidation via scavenging hydroxyl radicals and they attributed this activity to 3 dipeptides containing tyrosine residues49. Additionally, Kanbur et al.37 found that the antioxidant activity of RJ is not only due to its free radical scavenging activity but also due to the effects of RJ on the inhibition of enzymes that increase the peroxidation of endogenous lipids.
The antioxidant effect of RJ, which acts as a scavenger of free radicals, causes normal histological findings after treatment with RJ and NaF. Thus, RJ products can prevent and treat many liver dysfunctions since they are very rich in natural antioxidants. Additionally, the RJ products increase recovery from NaF-induced liver damage in a way that is partially reliant on their antioxidant qualities and bioavailability4.
In this study, the results showed that NaF induced a large amount of reactive oxygen species production accompanied by the decline of activities of antioxidant enzymes including SOD, CAT and GPx and elevation of MDA level. These findings illustrated the severe oxidative stress and damage and cellular toxicity induced by NaF and the ameliorative effects of RJ in the alleviation of hepatotoxicity and oxidative injury.
The obtained results clarified that NaF induced hepatic toxicity with the excessive triggering of reactive oxygen species that induced severe oxidative injury and histological alterations in the hepatic structure. These results are concurrent with obtaining normal biochemical levels in groups either treated with RJ or even combined with RJ and NaF. The RJ is considered an essential potential source of natural antioxidants capable of frustrating oxidative injury, which is considered the main cause of many current diseases.
Fluoride was determined to cause adverse effects on erythrocyte and liver tissue. Although several hypotheses have been proposed, the exact mechanism of fluoride toxicity has not been clearly defined. Thus, this present study provides the vision to understand the hepatoprotective effects of royal jelly against hepatotoxicity and oxidative stress induced by sodium fluoride in male rats. Since, royal jelly is a mixture of multi-components such as glucose, lipids, minerals, vitamins, acetylcholine and nucleic acids which are essential for the properties of royal jelly.
This project was spotted by the Deanship of Scientific Research (DSR), King Abdulaziz University, Jeddah. The author acknowledges with thanks DSR technical and support.